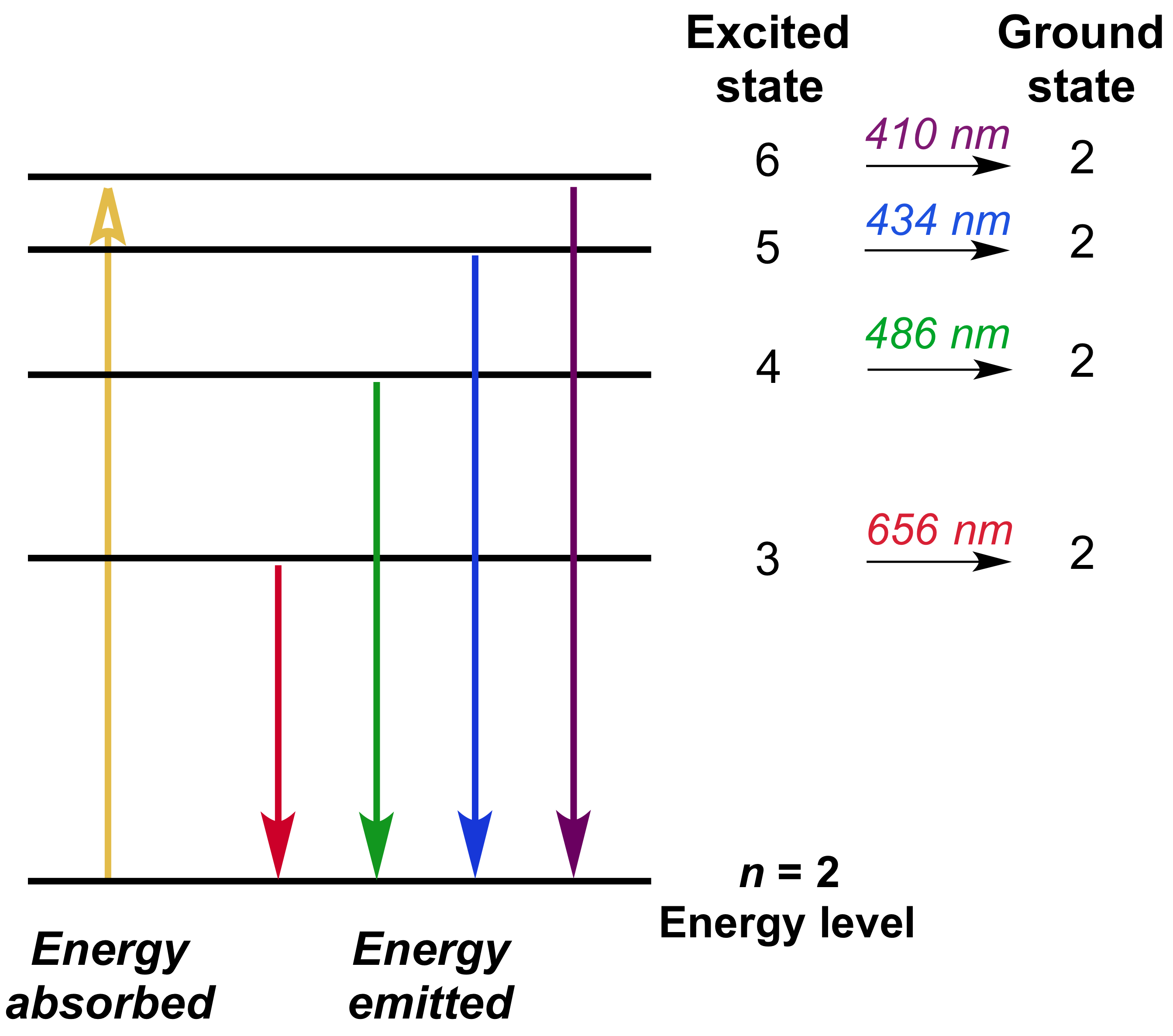
By using this method, we can forecast lines that we have not found yet. Explanation: Electrons move around the hydrogen atom in electron waves patterns. But Balmer did not understand why this formula would happen. The emission spectrum lines are the changes in the quantum energy levels of the single electron in the Hydrogen atom. Our eyes can not see the light in the ultraviolet region.Īt the end of the 19th century, it was already discovered that wavelengths appearing in the spectrum of hydrogen atoms are clearly classified.īalmer, a Swiss teacher, showed the position of the spectral line with a mathematical formula. There is one line in the red area, one line in the cyan area, some lines in the purple area, and many lines in the ultraviolet region. A formula is obtained which enables the electron concentration in hydrogen plasma to be determined by the last observable line with due account for outside (. It is possible to detect patterns of lines in both the ultraviolet and infrared regions of the spectrum as well. 
The hydrogen spectrum is complex, comprising more than the three lines visible to the naked eye. 
Electricity & Magnetism Toggle Child MenuĪt the time of Rutherford’s experiments, chemists analyzed chemical components using spectroscopy, and physicists tried to find what kind of order in complex spectral lines.įor example, a hydrogen arc tube containing hydrogen, a light element, shows a highly ordered spectrum compared with other elements. Hydrogen line profiles observed in the emission spectrum of a microwave discharge generated in a rectangular (TE 111 ) resonant cavity: a H at pressure. Generally, the emission spectrum of a chemical element or compound is the spectrum of frequencies of electromagnetic radiation. Emission lines ( discrete spectrum) Absorption spectrum with Absorption lines (discrete spectrum) Absorption lines for air, under indirect illumination, with the direct light source not visible, so that the gas is not there directly between source and detector. Extending hydrogen's emission spectrum into the UV and IR.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
